From Code to Control: The Therapeutic Potential of Epigenome Editing
Just as gene therapies are beginning to transform the treatment landscape for a wide range of diseases, a recent study introduces another powerful tool: epigenetic therapy. By directing a small complex of epigenetic editors to a specific target gene in mice, Cappelluti and colleagues successfully reduced the expression of PCSK9, a protein that regulates cholesterol. In this article, we will explore how epigenetic therapies came to be, how they work, and how they may offer new advantages over conventional gene therapies in treating complex diseases.
“Wow . . . what a bonanza for gene therapy!”
Inder M. Verma. Then editorial in Molecular Therapy, August 2000.
Barely a month after the announcement of the near-completion of the Human Genome Project—which aimed to map all the genes found in human DNA—researchers were already exploring ways to harness this wealth of information to cure genetic diseases1. We had a map; but we lacked the tools. Then, just 12 years later came the CRISPR revolution.
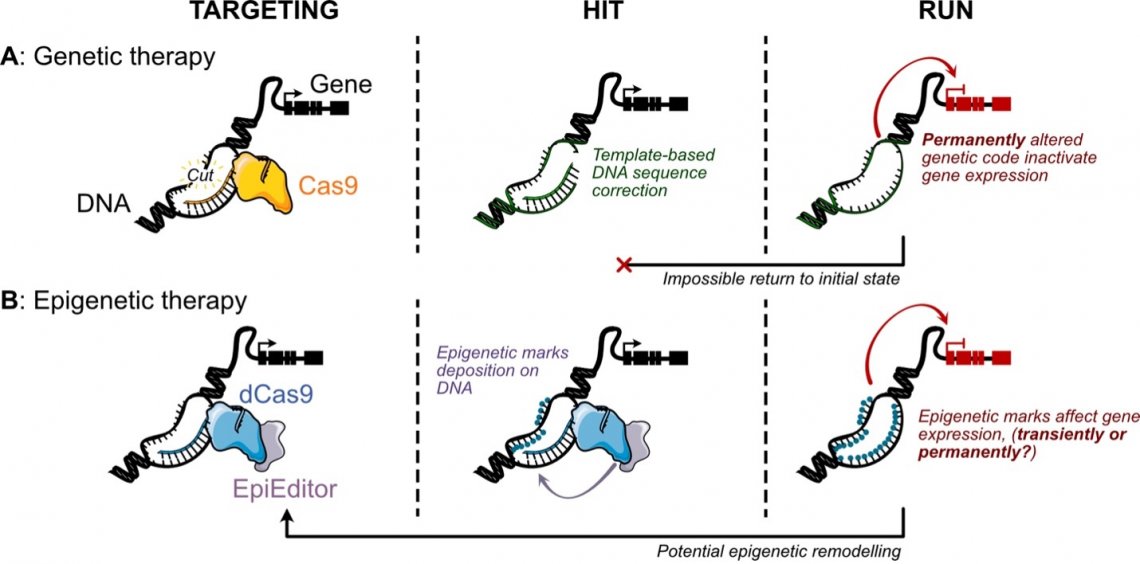
Derived from a bacterial “immune system,” the CRISPR/Cas9 editor protein (or Cas9) functions as molecular scissor, capable of precisely cutting DNA at specific, targetable sequences. This breakthrough enabled scientists to remove and correct mutated DNA sequences by providing a corrective template for mutant cells to repair their DNA (Figure 1A) 2. A mere year and a half after the gene editing capacities of CRISPR/Cas9 were published, CRISPR Therapeutics—founded by one of the pioneers of the Cas9 gene-editing strategy—raised over $80 million USD. Within two years, more than 150 patents for CRISPR-based applications were published 3. This fast-paced race would come to fruition in 2023 with the development of Casvegy by CRISPR Therapeutics, the first approved CRISPR-based gene therapy to treat well-known blood genetic disorders: sickle cell disease and β-thalassemia. This represents an incredible technological achievement, but remains limited in its impact. Indeed, the cost of this treatment is estimated at $2 million USD, and only a handful of patients across the US can be treated by the company per year 4. Despite these challenges, there are over 2000 potential gene therapies in development worldwide, and the market forecast is estimated to rise from 5 billion USD in 2022 to over 19 billion in 2027 5.
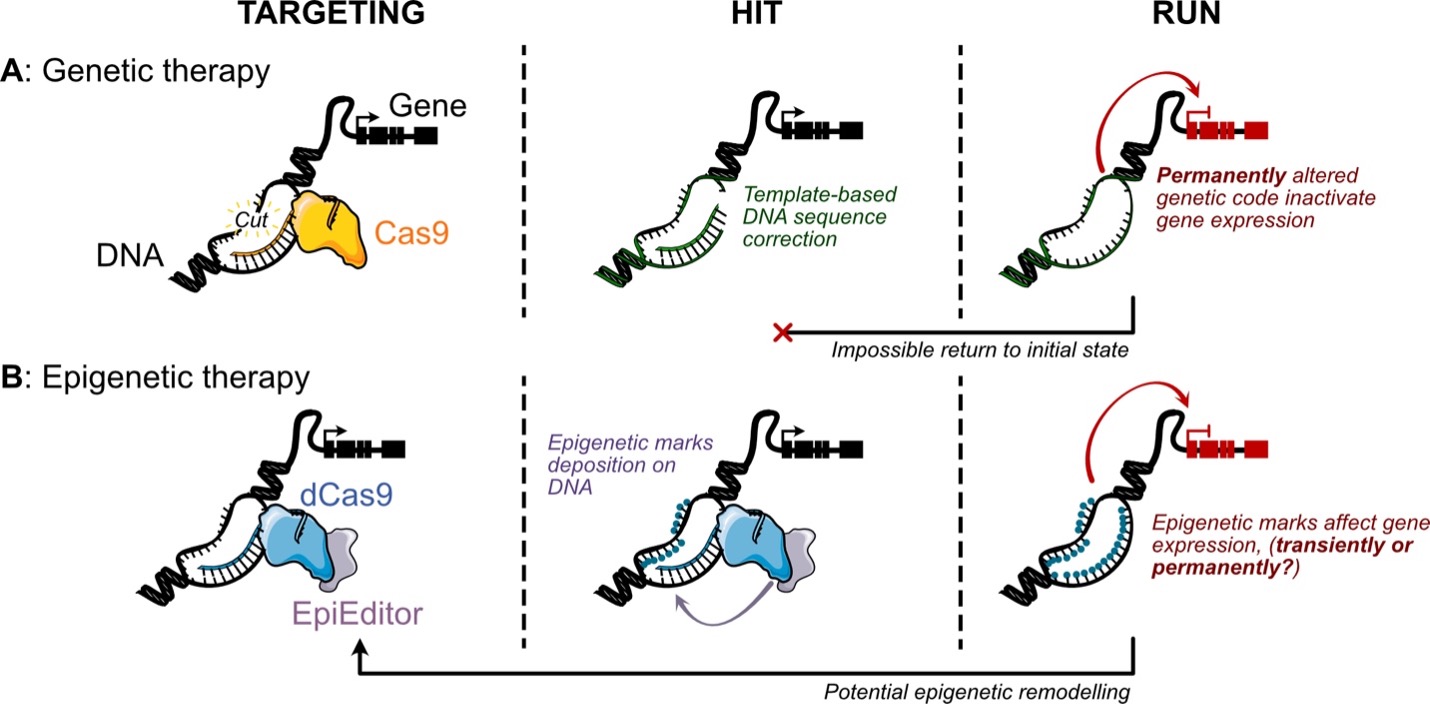
Yet, concerns regarding gene therapies persist. Firstly, our understanding of the precise role that genes play in diseases remains limited. Only a few conditions are known to be directly caused by single-gene mutations, and an estimated 60% of congenital abnormalities cannot currently be traced back to specific genetic factors. Notably, twin studies have estimated that less than 40% of diseases had a strong genetic linkage 6, but this number is disputed and could be overestimated 7. Indeed, it is now widely accepted that most diseases—including obesity, diabetes, cancer, and neurological disorders—result from a complex interplay of polygenic factors (involving multiple genes) and environmental influences. Another significant limitation of gene therapy is its irreversible nature. Any modifications introduced by the editor Cas9 are permanent, raising a host of ethical concerns and fears regarding persistent side effects. Off-target edits, in particular, represent a major risk (Figure 1A), as these unintended modifications can be difficult and costly to identify, and often impossible to correct. Simply put, once a mistake occurs, reversing it would be nearly impossible.
The promise of Epigenetic Therapies: from code to control.
Epigenetic marks are chemical modifications attached to DNA and the histone proteins that package it (see previous article: https://thisisepigenetics.ca/what-is-epigenetics). Epigenetic modifications can either enhance or repress the expression of certain genes. Unlike permanent genetic mutations, epigenetic modifications are dynamically added or removed by specialized enzymes and can respond rapidly to external factors such as stress, diet, and aging. Consequently, epigenetic marks are increasingly recognized as crucial to understanding how diseases emerge from the interaction between our genetic blueprint and environmental factors—addressing complexities that genetics alone cannot fully explain. In fact, epigenetic drugs are already widely in use, especially for cancer treatments 8. Building on the path paved by gene editing therapy, a recent study published in Nature provides groundbreaking evidence that epigenetic editing therapy could cure disease and address some of the primary concerns associated with gene editing therapy.
Research published by Martino Alfredo Cappelluti and Angelo Lombardo from the San Raffaele Scientific Institute in early 2024 demonstrates the potential of an epigenetic editing strategy to lower LDL—“bad” cholesterol levels—in mice 9. Their approach involves a combination of epigenetic editors—particularly those involved in DNA methylation—to reduce the expression of the PCSK9 gene specifically in the liver. They targeted the PCSK9 gene as it encodes a protein that controls the levels of blood circulating LDL. To direct these epigenetic editors to precise genomic locations, the researchers tested several targeting systems, including a dCas9 (“dead” Cas9) variant, which cannot cut DNA, and zinc-finger proteins (ZFPs), another class of DNA-binding proteins engineered to recognize specific sequences (Figure 1B).
A two-step guide for efficient epigenome editing: “Hit and Run”.
To achieve successful epigenetic editing, two major challenges must be addressed: (i) ensuring a precise “hit” (the correct targeting and deposition of the new epigenetic marks) and (ii) maintaining this modification over time (the “run”). These two elements are closely connected, as a stronger initial “hit” is thought to lead to a more durable effect.
To achieve robust repression of the PCSK9 gene, the researchers combined two types of repressive epigenetic editors: DNA methyltransferases, which chemically modify DNA, and histone methyltransferases, which alter histone proteins associated with DNA. They reasoned that this combination would yield a more pronounced and lasting silencing of PCSK9. Indeed, they were able to reduce PCSK9 levels by 38–52% for over 300 days following the injection of the epigenetic editor into mice’s liver. This long-term repression correlated with the establishment of a stable, repressive epigenetic domain at the targeted site near the PCSK9 gene, with minimal off-target effects 9.
Overall, these modifications led to a 30% reduction in LDL-cholesterol levels one month after treatment. To further test the durability of their approach, the researchers exploited the liver’s regenerative capacity after partial liver surgical removal (hepatectomy). Even after partial hepatectomy, mice treated with the epigenetic editors continued to show reduced PCSK9 expression, lower LDL-cholesterol levels, and displayed a persistent repressive epigenetic domain more than two months post-surgery 9.
Epigenetic Editing: A Future Pathway for Precision Medicine
This work lays strong foundations for epigenome editing as a viable therapeutic strategy. It achieved strong and durable repression of PCSK9 gene, resulting in a significant reduction of “bad” LDL-cholesterol with minimal off target effects and without any modification of the genetic code.
However, several challenges remain. While minimal off-target edits were reported, unwanted modifications still occurred. The dCas9 system showed greater precision than zinc-finger proteins (ZFPs), suggesting it may be more effective at minimizing these unintended changes. Another challenge is that the reversibility of epigenome editing has yet to be addressed, an aspect that could differentiate it from permanent genetic therapies.
It is also essential to consider the experimental conditions. These studies were conducted in mice under tightly controlled environmental parameters—factors from diet intake, to temperature, and even day length was all managed by researchers. Achieving similarly strong therapeutic effects in humans, who live in far more variable conditions, may prove more complex.
Nonetheless, the evidence is compelling: epigenetic edits can be introduced into a living organism, can persist over time, and can yield therapeutic benefits. Moreover, these tools expand the scope of possible therapeutic interventions for complex diseases. By shifting the focus from altering the genetic code to controlling its expression, epigenome editing and epigenetic drugs hold promise for slowing aging processes, mitigating biological degeneration, and treating complex conditions like cancer or addiction 8,10,11.
References.
1. Verma, I. M. & Gage, F. H. Genome and Gene Therapy. Molecular Therapy 2, 95 (2000).
2. Jinek, M. et al. A Programmable Dual-RNA–Guided DNA Endonuclease in Adaptive Bacterial Immunity. Science 337, 816–821 (2012).
3. Ledford, H. CRISPR, The Disruptor. Nature 522, 20–24 (2015).
4. Sheridan, C. The world’s first CRISPR therapy is approved: who will receive it? Nature Biotechnology 42, 3–4 (2023).
5. Bishen, S. & Doxzen, K. Why we need to accelerate global access to gene therapy. World Economic Forumhttps://www.weforum.org/stories/2022/10/gene-therapy-why-we-need-to-acc… (2022).
6. Polderman, T. J. C. et al. Meta-analysis of the heritability of human traits based on fifty years of twin studies. Nat Genet 47, 702–709 (2015).
7. Feldman, M. W. & Ramachandran, S. Missing compared to what? Revisiting heritability, genes and culture. Philosophical Transactions of the Royal Society B: Biological Sciences 373, 20170064 (2018).
8. Yoo, C. B. & Jones, P. A. Epigenetic therapy of cancer: past, present and future. Nat Rev Drug Discov 5, 37–50 (2006).
9. Cappelluti, M. A. et al. Durable and efficient gene silencing in vivo by hit-and-run epigenome editing. Nature(2024) doi:10.1038/s41586-024-07087-8.
10. Egger, G., Liang, G., Aparicio, A. & Jones, P. A. Epigenetics in human disease and prospects for epigenetic therapy. Nature 429, 457–463 (2004).
11. Janssen, S. Epigenetics and drug addiction: predisposition, chronic use, relapse and treatmentEpigenetics and drug addiction: predisposition, chronic use, relapse and treatment | This is Epigenetics. This is Epigeneticshttps://thisisepigenetics.ca/about-epigenetics/epigenetics-and-drug-add… (2023).
|
Learn more:
|