What is Epigenetics?
The Big Picture
Epigenetics is a rapidly growing field of scientific inquiry. It explains how our bodies have just one genetic code—the genome, written in DNA—but approximately 200 different types of cells carrying out a huge variety of functions, from the cells in our eyes that detect different types of light, to the cells in our intestines that specialize in absorbing nutrients, for example.
The term “epi-genetics” (literally “above genes”) was first coined in the 1950s by English embryologist Dr. Conrad Waddington to describe how a single cell develops into an embryo. Over time, the field evolved into the study of the variety of ways in which cells or organisms might acquire different features without changing the sequence of their genes. We now know that epigenetics is controlled by a collection of proteins and molecules that manipulate DNA to turn genes on and off, leading to different cell structures and functions, and we are becoming increasingly aware of the how epigenetics is involved in a range of diseases including cancer, autoimmune, respiratory, cardiovascular and neurodegenerative disorders.
In a nutshell, epigenetics is additional information layered on top of our genetic code—information that determines how that genetic code is used. It tells a liver cell to do liver things and a brain cell to do brain things, for example.
An analogy is the instruction manual used to manufacture a car. If it’s your job is to put on the tires, you don’t need information for windshield installation, seat upholstery or engine construction. Conveniently, the manual is broken into parts, chapters, sections and paragraphs so that you can focus solely on the information needed to put on the tires. The instruction manual is analogous to your genome—it is all of the information you need to make a human. The manual’s layout and organization, the epigenome—all of the biological chemistry that directs different cell types to read specific parts of the genome. In this way, working from the same information (your genome), your cells organize the information (using epigenetics) to work in concert, each playing their own role to form and maintain all of the tissues and organs of your body.
The Epigenome is Dynamic
The increasing number of references to epigenetics in many mainstream health and wellness practices has been fueled by accumulating evidence about the properties of the epigenome, and in particular about its dynamic nature; that is, that it can change in response to the environment. Evidence indicates, for example, that sleep, stress and diet can change the epigenome in ways that alter our cellular biological processes, with potential health implications. This “plasticity” of the epigenome means that some changes in cell, tissue or organ structure or function are not necessarily permanent. When things go awry with the epigenome resulting in disease, evidence indicates that there may be ways to correct it.
Considering our earlier analogy of instructions to make a car, epigenetic changes are like notes you’ve made in the margins of the manual: you might cross out some steps or add a few new ones, altering the way you read the instructions in the future. You might later change your mind on some of these notes and erase them. You might spill your coffee and leave a large stain on an important page. And if you were to photocopy your altered manual, the marks and stains would be copied along with it.
Epigenetic Changes Can be Passed on to Future Generations
Like the photocopied instruction manual, the epigenetic changes that occur throughout our lifetimes can be passed on, for better or for worse, to our children and even our grandchildren. Evidence for this, for example, comes from women who were pregnant during The Dutch Winter—a severe famine that occurred in the Netherlands at the end of the Second World War. Children who were in the womb during the famine grew up to experience significantly higher rates of diabetes and cardiovascular problems than those who were conceived after the famine. The epigenetic changes that occurred in the mothers were passed on, priming the children to better endure starvation but producing unwanted side-effects when nutrition became abundant.
Back to the Basics
Perhaps we’re getting ahead of ourselves, so let’s go back to the basics. If you already know a bit about DNA, feel free to scroll to the next section.
The Genetic Code
The genetic code, or DNA (deoxyribonucleic acid), within each of our cells is made up of four letters: A, T, C and G. Similar to the 1s and 0s of binary code, it is the order of these letters that carries the message. The letters stand for four molecules, called bases—Adenine, Thymine, Cytosine and Guanine. Within just one human cell, there are more than three billion of these bases strung together to form more than two metres of DNA.
So, what do the three billion bases of DNA code for? More than 21,000 different proteins. Why do we need 21,000 different proteins? Because proteins are the building blocks of life.
Proteins give our cells their shape and structure; they break down the molecules we eat and extract the energy; they replicate our genetic code so that our cells can divide; they hold our skin, organs and tissues together; they allow our hearts to beat, our muscles to contract and our immune cells to talk to one another; they carry oxygen throughout our bodies; they transmit signals from our toes to our brains. When it comes to the biological functions carried out by our bodies, proteins essentially do it all.
Each of our 21,000 different proteins are coded for by a small stretch of DNA called a gene. When a gene is read and used to make a protein, it is called gene expression. The process of reading the DNA code and making proteins occurs in three steps:
- First, the DNA is converted into an RNA (ribonucleic acid) code. RNA is similar to DNA, but it can be taken outside of the nucleus where it can be used to make protein.
- Once outside of the nucleus, the information coded by the RNA molecule is used to make a protein from building blocks known as amino acids.
- The protein then carries out its biological function either alone, or by binding to or working with other proteins.
This process has been given a fancy name: The Central Dogma of Biology.
Gene Expression
Each of the 200 different types of cells in our bodies have exactly the same genetic code, give or take a few bases. So how do they have such different characteristics?
Not all of our 21,000 proteins are needed by every cell all the time. To make so many unnecessary proteins would be a huge burden on our cells and an enormous waste of energy. To preserve the limited resources in our bodies, genes can be switched on and off. This is epigenetics—the information layered on top of our genetic code that determines which genes are expressed (switched on), and which are not (switched off). A brain cell does not need to carry out the same functions as a liver cell, for example, so it will turn off the genes required for liver functions and turn on the genes required for brain functions.
Histones
Before we dive deeper into how genes are switched on and off, we need to talk a bit about how DNA is structured.
Remember how we said that each of our cells contain more than two metres of DNA? Our cells range in size from 50 to 100 micrometres in diameter, with nuclei that are about five micrometres. A micrometre is one millionth of a metre; the width of a human hair is approximately 100 micrometres. Squishing two metres of DNA into a five-micrometre nucleus is like fitting a piece of string that is twice the length of California into a pea. In fact, if you took all the DNA in your body and put it end-to-end it would stretch 10 billion miles—all the way to Pluto and back.
To fit two metres of DNA into each of our cells, it is wound around packaging proteins called histones. The DNA strand is wrapped two and half times around each histone, forming something that looks like beads on a string. The DNA-protein hybrid, which is called chromatin, then condenses down further to form the 46 chromosomes that reside within the nucleus.
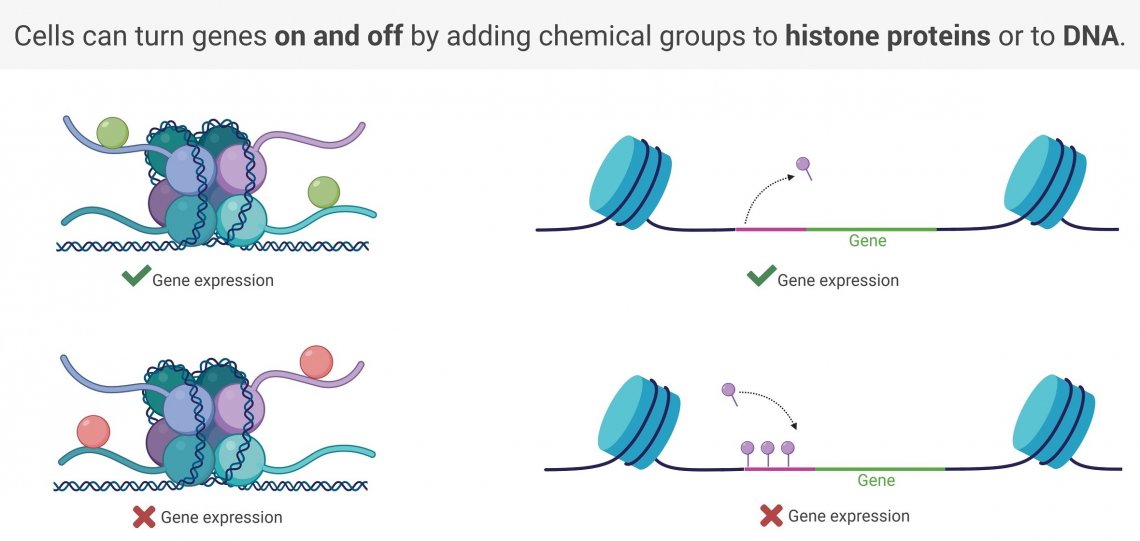
Histone Modification
For many years, scientists considered histone proteins to be nothing more than packaging material. But it turns out, they play another very important role—they are also “switches” that turn gene expression on and off.
The parts of the genetic code that are wrapped tightly around histone proteins cannot be read by our cells. For the central dogma of biology to work, the DNA-histone interactions must be loosened. This is the switch. Segments of DNA that are tightly wound around histone proteins are switched off, while those that are loosely wound are switched on.
So how does the switch get flipped? Our cells can tighten and loosen the DNA-histone interactions by chemically modifying the histone proteins. Adding on an activating chemical group causes the histone protein to become less “sticky”, loosening up the DNA and allowing for gene expression to occur. Inhibiting chemical groups increase the attraction between the DNA and the histone protein, tightening the interaction and preventing gene expression.
DNA Modifications
Like most biological processes, gene expression has multiple layers of regulation. In addition to histone modification, cells can turn off gene expression by chemically modifying the DNA itself.
At the beginning of every gene is a segment of DNA, called a promoter, which tells the cell where to start reading. Without a promoter, the cell won’t be able to find the beginning of the gene. By chemically modifying the DNA in these regions, adding on chemicals called methyl groups, the cells can “hide” their promoters, turning off gene expression. This type of switch is called DNA methylation. DNA methylation can be reversed to turn gene expression back on.