In order to grow, proliferate and spread throughout the body, rapidly dividing cancer cells require a constant stream of nutrients and access to the circulatory system. They accomplish this through a key hallmark of cancer—the formation of new blood vessels, which feed growing tumours the nutrients and oxygen needed to satiate their high metabolic requirements.
Tumours are large masses of cells with a high requirement for nutrients; without the formation of new blood vessels—a tightly regulated process called angiogenesis— cells within a tumour may experience limited nutrient access. Increased angiogenesis is also one of the mechanisms which grants tumours access to the circulatory system, which can then be used by the cancer cells to migrate through the body during metastasis.
Early in the conversion of healthy cells into cancer cells, angiogenesis within the tissue is increased. This occurs through the tight regulation of the many factors that either promote or prevent angiogenesis.
Regulation of angiogenesis
Normally, epigenetic marks ensure that most cells remain in an anti-angiogenic state, preventing excessive blood vessel formation with angiogenic genes being associated with both repressive and activating histone marks. In situations such as wound healing, when new blood vessels are required to repair damage, cells can rapidly shift to a pro-angiogenic state by removing the repressive histone marks. MicroRNA’s, small RNA molecules that regulate gene expression, have also been shown to regulate angiogenesis by modifying the expression of key angiogenic genes.
In angiogenesis, blood vessels are created by endothelial cells, but are attracted to tissues that release specific growth factors. The balance of angiogenesis within a tissue is partially regulated through exosomes. These membrane-bound extracellular vesicles can transfer transcription factors and other regulatory elements, including microRNA’s, between neighbouring cells within their local environment, coordinating their transcriptional activity. Exosomes offer a mechanism for tumours to alter the epigenetic environment of nearby cells by transferring epigenetic elements within their local environment.
Dysregulation of angiogenesis in cancer
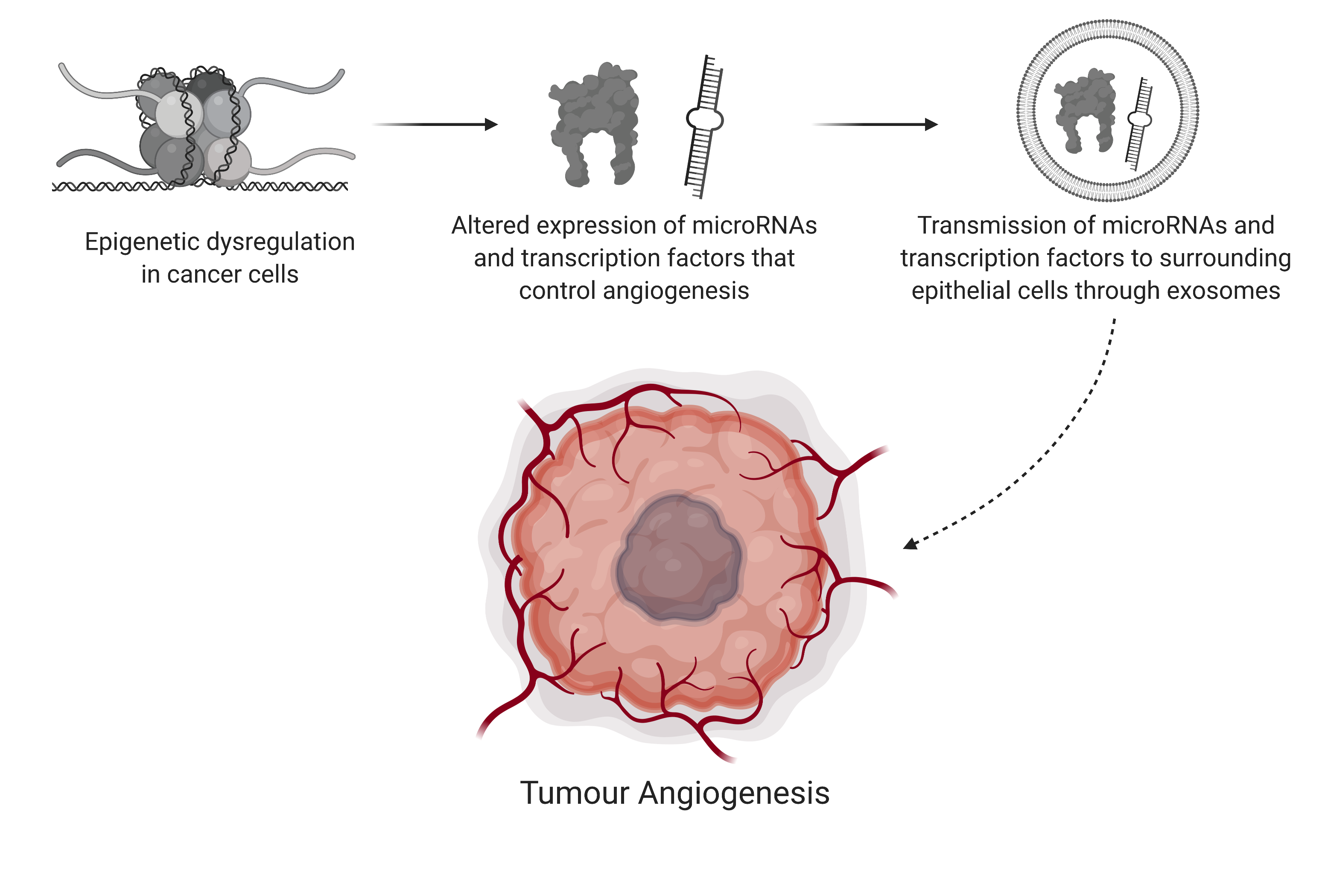
In some cancers, angiogenesis involves the gene erythroblast transformation-specific (ETS)-related gene (ERG). Increased expression of ERG has been shown to promote angiogenesis. To prevent unwanted blood vessel formation, ERG expression is usually turned off by the microRNA miR-200b-3p.
In hepatocellular carcinoma, however, this microRNA is downregulated, leading to the subsequent upregulation of ERG within the tumour mass itself. Interestingly, ERG expression was also found to be upregulated in the surrounding epithelial cells, leading to angiogenesis.
How do microRNA levels within cancer cells affect angiogenesis in surrounding epithelial cells? In a study published in Scientific Reports, researchers found the answer: exosomes. They found that exosomes produced by healthy liver tissue contained the microRNA which, when transferred to the surrounding epithelial cells, downregulated ERG and decreased angiogenesis. In contrast, the exosomes produced by cancerous liver cells contained less of the microRNA, leading to increased expression of ERG in the surrounding epithelial cells and increased angiogenesis.
These studies illustrate how regulation of gene expression through epigenetics, exosomes and microRNAs contribute to the formation of new blood vessels within tumours and their surrounding tissues. Enhancing the understanding of regulatory processes that are disrupted in cancer will aid in the discovery of novel therapeutics that may reduce new blood vessel formation, thereby starving tumours of essential nutrients and preventing their spread throughout the body.